New generation PROCESS CONTROL SYSTEM for Ethylene Oxide Sterilizers
Vacudyne’s new generation control is designed with user efficiency in mind. With an extremely user friendly Windows based graphical user interface, and intuitive drop down menus, Vacudyne’s new control system raises the bar for ease of use and efficiency.
A SQL database provides a secure environment to protect your vital data while allowing access to historical information. A Windows based Wonderware InTouch HMI displays current phase data in real time to keep you informed of cycle progress. End of cycle reports summarize the information in an easy to read format.
Add to that nearly 60 years of sterilization experience and with Vacudyne, you are truly partnering with an industry leader.
QUALIFIED STERILIZER OPERATOR TRAINING
Follow this link to our new Vacudyne Sterilizer Training Academy for details
Overview

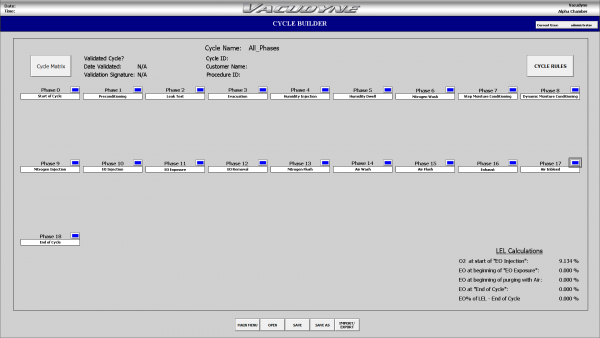
Cycle Building
- Flexable new cycle development capability by renaming and editing cycles reducing programming timing
- Extremely user friendly Windows based interface
- Drop down menu driven screens for flexible and efficient cycle development
- Rules based control prevents users from selecting phases out of sequence
-
Internal LEL calculation is designed to prevent user from creating an unsafe cycle
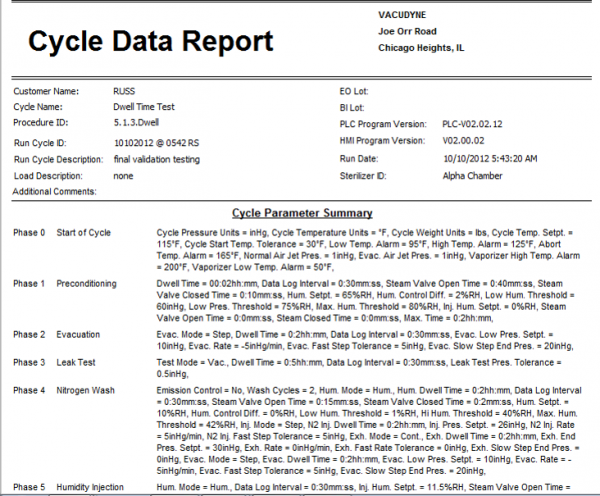
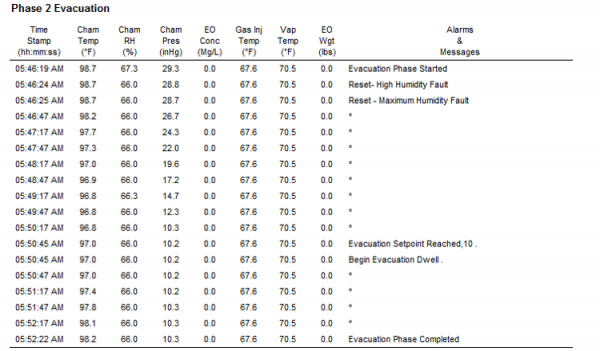
Reporting
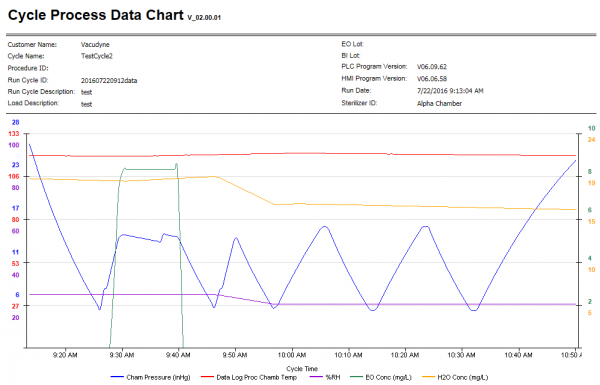
- Detailed cycle reports showing:
- Cycle Parameters
- Sterilization Process Data
- Alarms and Messages
- Graphs
- Optional reports for:
-
Summary Validation Report for Parametric
Release - Cycle to Cycle Comparison
- Product Temperature
-
Custom Designed to meet Customer
specifications
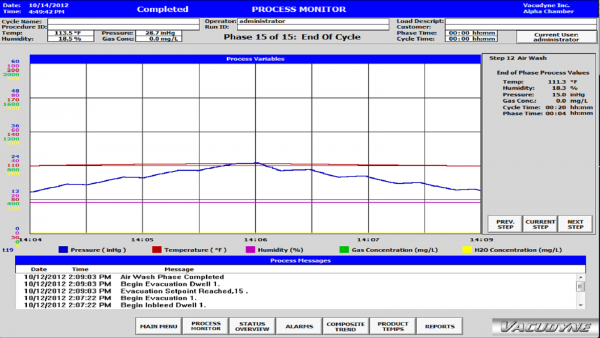
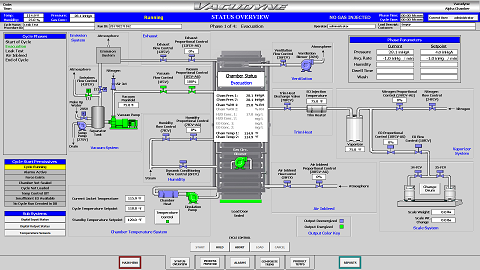
Process Monitoring
- The status of each Phase is monitored through the HMI with relevant data displayed on screen, for example: chamber pressure, chamber humidity, chamber gas concentration
- Messages and Alarms are tracked and displayed
- User has ability to "look back" at previous phases
Features
Features and Benefits
- User Friendly
- Powerful Report Package
- Flexible custom cycle development capability
- Multi Level Password Controlled Security
- Ease of Retrofitting with Older Systems
- Complete validation package
- 21 CFR part 11 Compliant
- Backed by over 55 years of experience
Applications
Available for both new installations and as a retrofit to replace existing Antares controls or other control systems.
Specifications
Control Hardware
- Allen-Bradley CompactLogix® PLC
- Windows based PC with AVENA InTouch HMI and Microsoft SQL database